« Back to all publications
Download this list in a RIS file or a BIB file or a PDF file
|
|
||||||||
Several aspects of ultrafast photochemistry in the condensed phase are discussed and illustrated by three examples from our laboratory. | ||||||||
|
 |
|||||||
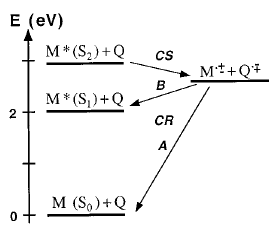
The dynamics of charge recombination (CR) of ion pairs formed upon electron-transfer quenching of Zn tetraphenylporphine (ZnTPP) in the S2Â state has been investigated by fluorescence upconversion. These ion pairs have two possible CR pathways:Â (A) a highly exergonic CR to the neutral ground state and (B) a moderately exergonic CR leading to the formation of ZnTPP in the S1Â state. Upon addition of quencher, the S2Â fluorescence decreases considerably, while the S1Â fluorescence is unaffected, indicating unambiguously that CR occurs via path B. A large fraction of the S2Â fluorescence quenching occurs in less than 100 fs. CR to the S1Â state of ZnTPP takes place with time constants around 400 fs. | ||||||||
|
||||||||
The dynamics of charge recombination (CR) of excited donorâacceptor complexes composed of methoxy-substituted benzenes and pyromellitic dianhydride were investigated in four different solvents using both the multiplex transient grating and the transient absorption techniques. At constant driving force, the CR dynamics are substantially faster than those with methyl-substituted benzenes as donors. In acetonitrile (ACN), the CR time constant decreases from 3.5 ps with anisole down to 240 fs with tetramethoxybenzene. In valeronitrile, the CR is always slower than in ACN but is, in most cases, faster than diffusional solvation. The free energy, the solvent, and the temperature dependence of the CR dynamics can be qualitatively well reproduced using the hybrid model of Barbara and co-workers after incorporation of the contribution of inertial motion to solvation. The ability of this model to account for the absence of normal region at small driving force is also examined. | ||||||||
|
|
Download this list in a RIS file or a BIB file or a PDF file
Contact:
Eric Vauthey
Physical Chemistry Department - Sciences II - University of Geneva
30, Quai Ernest Ansermet - CH-1211 Geneva 4 (Switzerland)
© All rights reserved by Eric Vauthey and the University of Geneva
Design and code by Guillaume Duvanel




