A bacterium that attacks burn victims will soon be unarmed
A study from UNIGE reveals the strategies adopted by a bacterium that proliferates in the wounds of burn victims and threatens them with sepsis. It opens the way to new therapies.
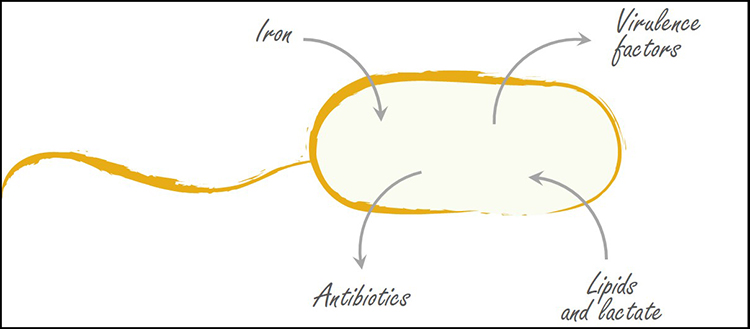
Overview of the mechanisms implemented by Pseudomonas aeruginosa in patient exudates. (© UNIGE)
The bacterium Pseudomonas aeruginosa is one of the main causes of infections and sepsis in people suffering from severe burns because it is difficult, if not impossible, to fight. Researchers at the University of Geneva (UNIGE), Switzerland, have succeeded in revealing the dynamics of the pathogen’s physiology and metabolism during its growth in exudates, the biological fluids that seep out of burn wounds. Published in the journal Frontiers in Cellular and Infection Microbiology, this study allows to follow step-by-step the strategies developed by Pseudomonas aeruginosa to proliferate and, thus, to guide the development of innovative treatments to counter them.
Pseudomonas aeruginosa has recently been classified by the World Health Organization as one of the priority pathogens to be targeted for the development of new treatments. This dangerous bacterium is responsible for various types of potentially lethal acute or chronic infections. Patients suffering from severe burns constitute a high-risk group, as this agent develops in wounds and adapts to its environment to proliferate and colonize the host, which can cause sepsis.
This micro-organism, widely present in the environment, takes advantage of a weakening of the host to set up various mechanisms that facilitate its multiplication. “We wanted to know what weapons were produced and used by P. aeruginosa and when during the infection. We therefore identified the genes that were expressed by bacteria that came into contact with patients’ exudates and developed in them”, explains Karl Perron, director of the Bacteriology Laboratory at the Department of Botany and Plant Biology of the UNIGE Faculty of Sciences.
The expression analysis of the some 6’000 bacterial genes was coupled with the analysis of the compounds used by P. aeruginosa, in collaboration with researchers from the Lausanne University Hospital (CHUV) and the Bern University Hospital.
A pathogen exposed
“The bacterium very rapidly overexpresses all the genes coding for proteins that make it possible to capture the host’s iron, because it is necessary for its growth and proliferation”, explains Manuel Gonzalez, a member of the Geneva team and first author of the article. These include both siderophores, molecules binding to soluble iron, and hemophores, real “molecular vacuum cleaners” that extract iron bound to red blood cells. At the same time, the bacterium also produces various systems for exporting and importing these molecules, before and after they have captured iron.
The researchers also found that P. aeruginosa uses mainly lactate, lipids and collagen from the host as a source of nutrients. In order to access these compounds, the bacterium rapidly produces and secretes specific enzymes capable of destroying the surrounding tissue.
“The biologists have shown that these virulence factors are produced in exudates even when the latter contain relatively small quantities of P. aeruginosa, suggesting that this pathogen rapidly becomes aggressive in burn wounds. We also observed that it generates pumps to expel molecules, which could allow it to resist to certain antibiotics”, adds Karl Perron.
Introducing a Trojan horse into the colony
These results, which reveal the dynamics of the physiology and metabolism of P. aeruginosa in a burn context, provide crucial elements for the development of new therapeutic approaches. “Since the availability of iron is a limiting factor for bacterial growth, a Trojan horse type strategy should be considered, which is under development. This consists in binding certain antibiotics to siderophores so that the bacterium imports them in large quantities, together with soluble iron”, explains Manuel Gonzalez. Molecules inhibiting specific virulence factors could also be administered at an early stage.
For this research, microbiologists have used an alternative method to animal experimentation. The analysis of exudates, their use and the synthesis of an artificial medium mimicking this particular environment have earned Karl Perron the ‘UNIGE 3R 2017 Prize’. The 3Rs rule (reduce, replace, refine) has established itself internationally as the foundation of the ethical approach applied to animal experimentation, and is actively promoted by UNIGE. It aims at reducing the number of animals used, refining experimental conditions to improve animal welfare, and replacing animal models with other experimental methods when possible.
27 Feb 2018
