Research
Eukaryotic cells contain a large number of different lipid species. In fact, the number of different lipid species probably exceeds the number of genes in many organisms. The main goal of our research is to understand the functions of membrane lipids in cell biology and physiology. To this end, we need to develop a comprehensive understanding about lipid distribution and homeostasis in cells and how these are regulated.
We are using a wide variety of approaches to reach our goal, including genetics, biochemistry, cell biology, chemical biology, synthetic biology, and analytical approaches including mass spectrometry. We are also using a number of experimental systems, with emphasis on the yeast S. cerevisiae, but including C. elegans, and mammalian tissue culture cells. Although our studies currently lead us mainly to study the functions of glycosylphosphatidylinositols, sterols and sphingolipids, we also work on other lipid species when they are implicated in function through our unbiased approaches.
Genetic Control of Lipid Homeostasis and Function
In order to understand the genetic control of lipid metabolism and correlate the quantity of particular lipids with cellular functions we have begun to compare the lipid profiles of a number of yeast mutants, including those defective in the regulation of protein phosphorylation and those representing proteins present in the early secretory pathway, the site of most lipid synthesis. We have created a lipidomics platform in the French part of Switzerland with facilities at the EPFL in Lausanne and in the Biochemistry department at the University of Geneva. We use the Orbitrap LTQ (Thermo) for high resolution mass spectrometry and profiling and the triple quadrupole TSQ Vantage (Thermo) for routine targeted quantification. All machine are equipped with a Nanomate (Advion) for nanospray. In this way, we can compare large number of mutants. Sterols are analysed by GC-MS using a 320-MS (Varian). This project is part of a SystemsX.ch project, LipidX.ch. We collaborate with the Wenk group at the National University of Singapore, with whom we have previously shown that modification of sterol biosynthesis causes changes in sphingolipid composition. Using genetics we could show that sterols and sphingolipids function together to carry out a large number of biological processes.We are also developing similar techniques for siRNA-treated mammalian tissue culture cells and collaborate with the group of Taroh Kinoshita on GPI mutants, with Kai Johnsson on the effects of drugs on lipid composition. We have interesting new insights into the role of lipids in a mouse disease model in collaboration with Bernard Thorens.
Metabolic Engineering to Study Lipid Function
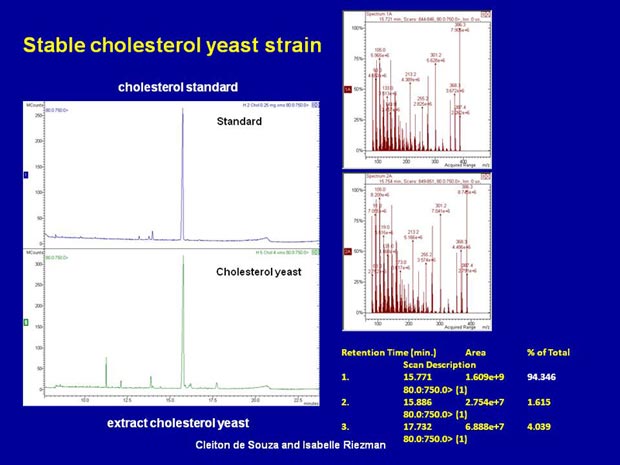
Previously we have studied the role of sterols in diverse cellular processes by introducing mutations in the ergosterol biosynthesis pathway into yeast. This yields strains with a variety of unnatural sterols. In a complementary approach we have engineered yeast strains to produce natural sterols, such as cholesterol or campesterol, which are present in other organisms where they fulfill sterol functions. Besides providing a useful host for expressing sterol-dependent membrane proteins this approach should allow us to differentiate between functions based on the physical versus structural properties of the sterol.We are also producing strains with altered sphingolipids, in particular, with different acyl chain lengths. This approach allows us to address the importance of acyl chain length in sphingolipid biology, but also to ncover new sphingolipid functions in endocytosis and cell division.
GPI-anchored Protein Biosynthesis and Transport
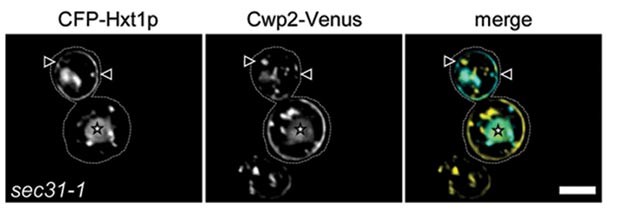
Glycosylphosphatidylinositol (GPI) anchored proteins are a class of membrane anchored proteins that are not exposed to the cytoplasm and therefore pose particular problems for protein sorting in the secretory pathway. They are processed post-translationally to attach a completed GPI to a freshly generated carboxyterminus by the GPI transamidase. Using biochemical assays we showed that GPI-anchored proteins are sorted from other secretory proteins upon ER exit. Recently, we have been able to visualize this sorting event in vivo and we have found that GPI-anchor remodeling is necessary for GPI-anchored protein concentration in ER exit sites. We are currently studying the mechanism of GPI-anchored protein sorting and concentration and the role of the p24 family of proteins in GPI-anchored protein traffic.
Roles of Ceramides, Sphingolipids and Glycolipids in C. elegans
We have recently shown, together with the group of Jean-Claude Martinou, that mutations in ceramide synthases affect the ability of worms to survive certain stresses, in particular, anoxia.This ability correlates with the amount of sphingolipids with specific chain lengths. We are currently attempting to determine how sphingolipids affect survival.In collaboration with Giovanni Lesa’s lab we are also examining mutants in glycolipid biosynthesis. Both of these projects have required setting up mass spectrometry tools to quantify lipids from C. elegans using high resolution mass spectrometry.