- Zhao, Y.; Beuchat, C.; Domoto, Y.; Gajewy, J.; Wilson, A.; Mareda, J.; Sakai, N.; Matile, S. “Anion-π Catalysis” J. Am. Chem. Soc. 2014, 136, 2101-2111
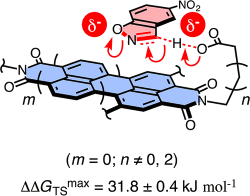
The introduction of new noncovalent interactions to build functional systems is of fundamental importance. We here report experimental and theoretical evidence that anion−π interactions can contribute to catalysis. The Kemp elimination is used as a classical tool to discover conceptually innovative catalysts for reactions with anionic transition states. For anion−π catalysis, a carboxylate base and a solubilizer are covalently attached to the π-acidic surface of naphthalenediimides. On these π-acidic surfaces, transition-state stabilizations up to ΔΔGTS = 31.8 ± 0.4 kJ mol–1 are found. This value corresponds to a transition-state recognition of KTS = 2.7 ± 0.5 μM and a catalytic proficiency of 3.8 × 105 M–1. Significantly increasing transition-state stabilization with increasing π-acidity of the catalyst, observed for two separate series, demonstrates the existence of “anion−π catalysis.” In sharp contrast, increasing π-acidity of the best naphthalenediimide catalysts does not influence the more than 12 000-times weaker substrate recognition (KM = 34.5 ± 1.6 μM). Together with the disappearance of Michaelis–Menten kinetics on the expanded π-surfaces of perylenediimides, this finding supports that contributions from π–π interactions are not very important for anion−π catalysis. The linker between the π-acidic surface and the carboxylate base strongly influences activity. Insufficient length and flexibility cause incompatibility with saturation kinetics. Moreover, preorganizing linkers do not improve catalysis much, suggesting that the ideal positioning of the carboxylate base on the π-acidic surface is achieved by intramolecular anion−π interactions rather than by an optimized structure of the linker. Computational simulations are in excellent agreement with experimental results. They confirm, inter alia, that the stabilization of the anionic transition states (but not the neutral ground states) increases with the π-acidity of the catalysts, i.e., the existence of anion−π catalysis. Preliminary results on the general significance of anion−π catalysis beyond the Kemp elimination are briefly discussed.
DOI: 10.1021/ja412290r
open archive unige:34124 • pdf ![]()
