- Benz, S.; Macchione, M.; Verolet, Q.; Mareda, J.; Sakai, N.; Matile, S. “Anion Transport with Chalcogen Bonds” J. Am. Chem. Soc. 2016, 138, 9093-9096
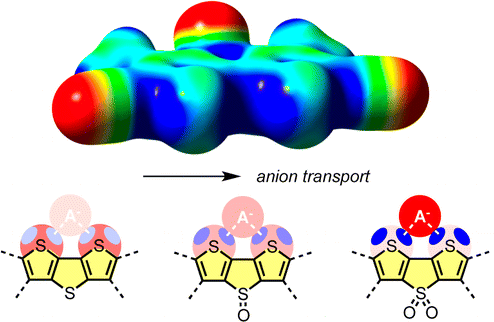
In this report, we introduce synthetic anion transporters that operate with chalcogen bonds. Electron-deficient dithieno[3,2-b;2′,3′-d]thiophenes (DTTs) are identified as ideal to bind anions in the focal point of the σ holes on the cofacial endocyclic sulfur atoms. Anion binding in solution and anion transport across lipid bilayers are found to increase with the depth of the σ holes of the DTT anionophores. These results introduce DTTs and related architectures as a privileged motif to engineer chalcogen bonds into functional systems, complementary in scope to classics such as 2,2′-bipyrroles or 2,2′-bipyridines that operate with hydrogen bonds and lone pairs, respectively.
DOI: 10.1021/jacs.6b05779
open archive unige:85614 • pdf ![]()
