Liste
Précédente Suivante
Publication 185
-
P. Müller, D. Riegert,
“Desymmetrization of spiroactivated meso-cyclopropanes via nucleophilic substitution”
Tetrahedron 2005, 61, 4373-4379.
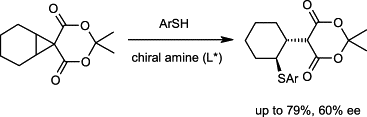
The spiro-activated cyclopropane 1 undergoes desymmetrization either with Li-thiophenoxide in the presence of a chiral complexing ligand, or with ion pairs formed from thiophenols and aromatic chiral amines. The latter procedure is more efficient and provides the ring-opened thioether 6 in up to 79% yield and up to 60% ee.
DOI : 10.1016/j.tet.2005.03.005
archive ouverte unige:6972
