Publication 34
- Acetylene Derivatives of Cationic Diazaoxatriangulenes and Diaza [4]Helicenes – Access to Red Emitters and Planar Chiral Stereochemical Traits
Ondrisek, P.; Elie, M.; Pupier, M.; de Aguirre, A.; Poblador Bahamonde, A. I.; Besnard, C.; Lacour, J.
Chem. Eur. J. 2022, 28, e202104405
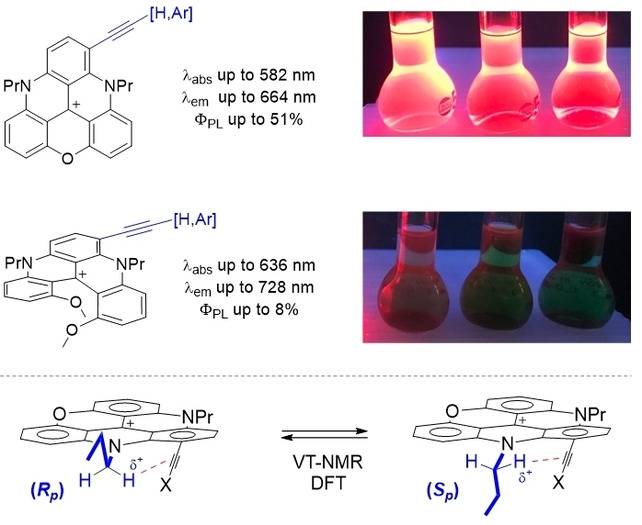
Cationic triangulenes, and related helicenes, constitute a rich class of dyes and fluorophores, usually absorbing and emitting light at low energy, in the orange to red domains. Recently, to broaden the scope of applications, regioselective late-stage functionalizations on these core moieties have been developed. For instance, with the introduction of electron-donating groups (EDGs), important bathochromic shifts are observed pushing absorptions towards or in the near-infrared (NIR) spectral domain while emissive properties disappear essentially completely. Herein, to upset this drawback, acetylene derivatives of cationic diazaoxa triangulenes (DAOTA) and [4]helicenes are prepared (16 examples). Contrary to other EDG-functionalized derivatives, C≡C-functionalized products remain broadly fluorescent, with red-shifted absorptions (Δλabs up to 25 nm) and emissions (Δλem up to 73 nm, ΦPL up to 51%). Quite interestingly, a general dynamic stereoisomerism phenomenon is evidenced for the compounds derived from achiral DAOTA cores. At low temperature in 1H-NMR spectroscopy (218 K), N-CH2 protons become diastereotopic with chemical shifts differences (Δδ) as high as +1.64 ppm. The signal coalescence occurs around 273 K with a barrier of ~12 kcal.mol-1. This phenomenon is due to planar chiral conformations (Sp and Rp configurations), induced by the geometry of the alkyl (n-propyl) side-chains next to the acetylenic substituents. Finally, DFT calculations offer a valuable insight on geometries, stereodynamics and on the large difference in NMR for some of the diastereotopic protons.
DOI : 10.1002/chem.202104405
archive ouverte unige:159531
