Publication 37
- Chemo- and Regioselective Multiple C(sp2)−H Insertions of Malonate Metal Carbenes for Late-Stage Functionalizations of Azahelicenes
Nikolova, Y.; Fabri, B.; Moneva Lorente, P.; Guarnieri-Ibáñez, A.; de Aguirre, A.; Soda, Y.; Pescitelli, G.; Zinna, F.; Besnard, C.; Guénée, L.; Moreau, D.; Di Bari, L.; Bakker, E.; Poblador Bahamonde, A. I.; Lacour, J.
Angew. Chem. Int. Ed. 2022, 61, e202210798
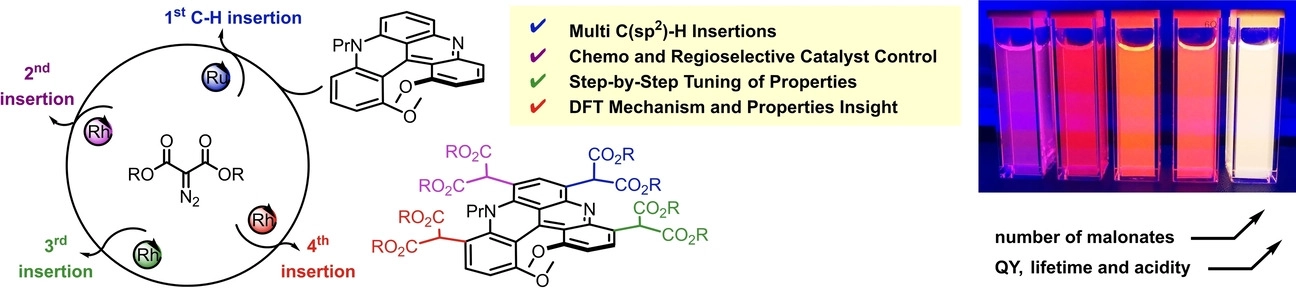
Chiral quinacridines react up to four times, step-by-step, with α-diazomalonates under Ru(II) and Rh(II) catalysis. By selecting the catalyst, [CpRu(CH3CN)3][PF6] (Cp = cyclopentadienyl) or Rh2(oct)4, chemo and regioselective insertions of derived metal carbenes are achieved in favor of mono- or bis-functionalized malonate derivatives respectively (r.r. > 49:1, up to 77% yield, 12 examples). This multi-introduction of malonate groups is particularly useful to tune optical and chemical properties such as absorption, emission or Brønsted acidity but also cellular bioimaging. Density-functional theory further elucidates the origin of the carbene insertion selectivity and also showcases the importance of conformations in the optical response.
DOI : 10.1002/anie.202210798
archive ouverte unige:164073
