Publication 101
- “The Photohydration of N-Alkylpyridinium Salts: Theory and Experiment”
King, R. A.; Lüthi, H. P.; Schaefer III, H. F.; Glarner, F.; Burger, U.
Chem. Eur. J. 2001, 7, 1734-1742.
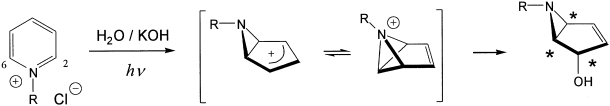
Bicyclic aziridines formed by the irradiation of pyridinium salts in basic solution have recently been recognized to have great synthetic potential. We have undertaken a joint computational and experimental investigation of the mechanism of this photoreaction. We have computationally determined the structures and relative energies of the relevant stationary points on the lowest potential energy surface (PES) of the pyridinium and methylpyridinium ions. Two important intermediates are shown to be bound minima on the ground-state PES: azoniabenzvalene and a 6-aza[3.1.0]bicyclic ion with an exo-oriented substituent (analogous to prefulvene). We advance a mechanism which involves initial formation of this exo-bicyclic ion, followed by nitrogen migration around the ring via the azoniabenzvalene intermediate. Thus, the barrier separating the two intermediates is the factor that determines the degree of scrambling observed in the photoproducts when the carbon atoms are labeled with deuterium or substituted with additional methyl groups. For N-methylpyridinium, the exo-methyl bicyclic ion was computed to be ≈ 1 kcal mol-1 lower in energy than N-methyl-azoniabenzvalene. The transition state was computed to lie several kcal mol-1 above the exo-methyl bicyclic ion (+8.4 kcal mol-1, 6-31G* RHF; +3.7 kcal mol-1, 6-31G* B3LYP), but still well below the energy available from the 254 nm excitation of the N-methylpyridinium ion. The computed relative energies correspond splendidly with several experimental findings which include the preference for exo products, the results of deuterium labeling, and the impact of additional substituent methyl groups on the product distribution.
DOI : 10.1002/1521-3765(20010417)7:8<1734::AID-CHEM17340>3.0.CO;2-0
archive ouverte unige:7768
