-
Conformational Dynamics of Single G Protein-Coupled Receptors in Solution
Samuel Bockenhauer1, Alexandre Fürstenberg1, Xiao Jie Yao, Brian K. Kobilka, and W. E. Moerner
J. Phys. Chem. B 2011, 115(45), 13328-13338
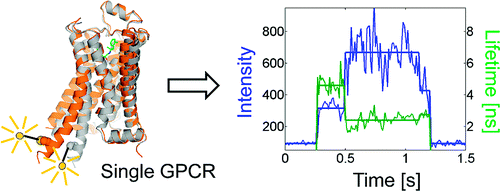 |
G protein-coupled receptors (GPCRs) comprise a large family of seven-helix transmembrane proteins which regulate cellular signaling by sensing light, ligands, and binding proteins. The GPCR activation process, however, is not a simple on–off switch; current models suggest a complex conformational landscape in which the active, signaling state includes multiple conformations with similar downstream activity. The present study probes the conformational dynamics of single β2-adrenergic receptors (β2ARs) in the solution phase by Anti-Brownian ELectrokinetic (ABEL) trapping. The ABEL trap uses fast electrokinetic feedback in a microfluidic configuration to allow direct observation of a single fluorescently labeled β2AR for hundreds of milliseconds to seconds. By choosing a reporter dye and labeling site sensitive to ligand binding, we observe a diversity of discrete fluorescence intensity and lifetime levels in single β2ARs, indicating a varying radiative lifetime and a range of discrete conformational states with dwell times of hundreds of milliseconds. We find that the binding of agonist increases the dwell times of these states, and furthermore, we observe millisecond fluctuations within states. The intensity autocorrelations of these faster fluctuations are well-described by stretched exponential functions with a stretching exponent β ∼ 0.5, suggesting protein dynamics over a range of time scales.
DOI: 10.1021/jp204843r
