Liste
Précédente Suivante
Publication 141
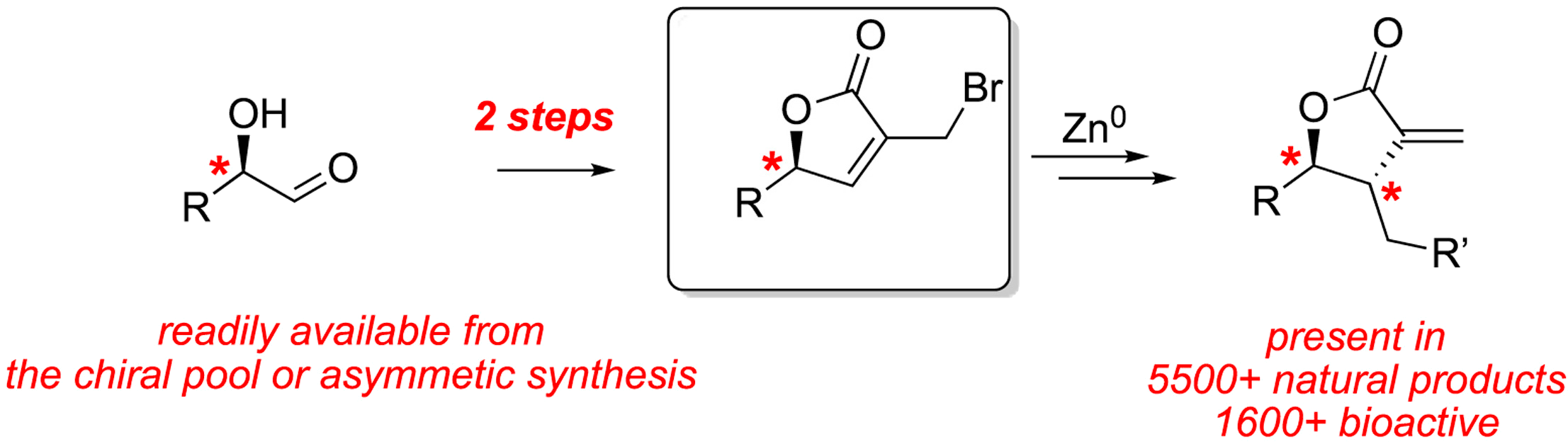
- Rapid and scalable synthesis of chiral bromolactones as precursors to α-exo-methylene-γ-butyrolactone-containing sesquiterpene lactones
R. Lagoutte, M. Pastor, M. Berthet, N. Winssinger
Tetrahedron 2018, 74, 6012-6021

The sesquiterpene lactones cover a diverse and pharmacologically important diversity space. In particular, the electrophilic α-exo-methylene-γ-butyrolactone moiety that is preponderant in this natural product family has been shown to readily engage in covalent inhibition via conjugate addition of cysteine residues in target proteins. However, the synthetic accessibility of sesquiterpenes or related probes to investigate their mode of action remains laborious. Herein, we present a rapid and scalable route to chiral bromolactones as enabling precursors in the synthesis of sesquiterpene lactones.
DOI : 10.1016/j.tet.2018.08.045
archive ouverte unige:107781
