Publication 92
- Expanding the Scope of PNA-Encoded Synthesis (PES): Mtt-Protected PNA Fully Orthogonal to Fmoc Chemistry and a Broad Array of Robust Diversity-Generating Reactions
D. Chouikhi, M. Ciobanu, C. Zambaldo, V. Duplan, S. Barluenga, N. Winssinger
Chem. Eur. J. 2012, 18, 12698-12704
[ Corrigendum: Chem. Eur. J. 2013, 19, 8022]
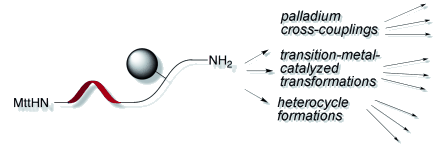
Nucleic acid-encoded libraries are emerging as an attractive and highly miniaturized format for the rapid identification of protein ligands. An important criterion in the synthesis of nucleic acid encoded libraries is the scope of reactions that can be used to introduce molecular diversity and devise divergent pathways for diversity-oriented synthesis (DOS). To date, the protecting group strategies that have been used in peptide nucleic acid (PNA) encoded synthesis (PES) have limited the choice of reactions used in the library synthesis to just a few prototypes. Herein, we describe the preparation of PNA monomers with a protecting group combination (Mtt/Boc) that is orthogonal to Fmoc-based synthesis and compatible with a large palette of reactions that have been productively used in DOS (palladium cross-couplings, metathesis, reductive amination, amidation, heterocycle formation, nucleophilic addition, conjugate additions, Pictet–Spengler cyclization). We incorporate γ-modifications in the PNA backbone that are known to enhance hybridization and solubility. We demonstrate the robustness of this strategy with a library synthesis that is characterized by MALDI MS analysis at every step.
DOI : 10.1002/chem.201201337
archive ouverte unige:24667
