Importance of heteroaggregation in aquatic systems
Article about Heteroaggregation of CeO2 nanoparticles in presence of alginate and iron (III) oxide published in Science of The Total Environment.
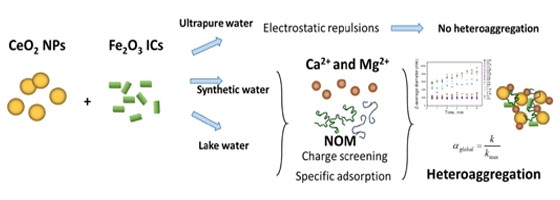
When manufactured nanoparticles are released to natural waters, heteroaggregation between nanoparticles and water compounds is expected to occur and play a key role in nanoparticle fate, transport and transformation. In this work, the heteroaggregation between CeO2 nanoparticles and Fe2O3 inorganic colloids, which represent the main inorganic fraction from Lake Geneva water, is studied. The heteroaggregation processes between CeO2, Fe2O3 and alginate in multiple water samples are investigated using zeta potential and z-average diameter measurements. The kinetics of heteroaggregation of individual components as well as mixtures of CeO2 nanoparticles and Fe2O3 colloids and alginate are studied using time resolved dynamic light scattering. The global attachment efficiency (αglobal) is calculated using data from kinetic experiments. αglobal for pristine CeO2 nanoparticles varied from 0.5 to 0.7 in lake and synthetic waters and is found around 1 for pristine Fe2O3 and mixture CeO2 and Fe2O3. Our findings demonstrate that heteroaggregation is highly dependent on environmental conditions and resulting electrostatic scenarios. No heteroaggregation at pH 8 between CeO2, Fe2O3 and alginate is observed in ultrapure water, because of electrostatic repulsions between negatively charged compounds. In synthetic and lake waters, the situation is opposite. Indeed, specific adsorption of divalent cations and presence of salt are found to promote heteroaggregation via cation bridging and screening effects. The kinetic experiments indicate that aggregation rate of pristine Fe2O3 is higher (89 nm/min in lake water) compared to pristine CeO2 nanoparticles (50 nm/min) and on the same level as mixture of CeO2 and Fe2O3 (96 nm/min). Low alginate concentration, 0.25 mg/L, has no effect on heteroaggregation in mixture of CeO2 and Fe2O3 in lake and synthetic waters. On the other hand, in natural water, the presence of higher alginate concentration, 2 mg/L, is found to reduce the heteroaggregation rate.
Oriekhova, Olena, and Serge Stoll. 2019. ‘Heteroaggregation of CeO2 Nanoparticles in Presence of Alginate and Iron (III) Oxide’. Science of The Total Environment 648 (January): 1171–78. https://doi.org/10.1016/j.scitotenv.2018.08.176.
A personalized URL providing 50 days' free access to this article:
Anyone clicking on this link before October 13, 2018 will be taken directly to the final version of the article on ScienceDirect, which they are welcome to read or download. No sign up, registration or fees are required.
Version deposited in Archive ouverte UNIGE click here
30 Aug 2018